News

Welcome to AIHTA!
AIHTA is an academic non-profit institute. At our website we wish to present our research and give support for health care decision-making. Here all of our reports are available for free. For further information please contact the AIHTA team.
In the News section you will find our most recent publications: newsletter, project-reports etc. Previous reports you might find via Research areas, Research projects and Repository.
Announcement
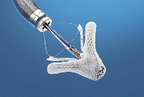
Transcatheter implantable devices for mitral valve repair in adults with chronic mitral valve regurgitation (Mitraclip®, Carillon®, NeoChord DS1000)
Mitral valve regurgitation (MR) or mitral insufficiency is a common disease esp. among the elderly. MR may be acute or chronic, depending on the underlying pathology: accordingly, it must be distinguished between primary (degenerative) MR (DMR) and secondary (functional) MR (FMR). Since recent years new minimally invasive procedures are available for inoperable or high-risk patients. This assessment examines the question of efficacy and safety of mitral valve repair using minimally invasive procedures such Mitraclip® for inoperable patients with DMR or FMR; Carillon® inoperable patients with FMR; NeoChord DS 1000 for operable patients with DMR to answer.
Two of the evaluated medical devices, NeoChord DS1000 and CARILLON® are still at an early stage of development. The MitraClip® however, was already implanted worldwide in around 23,000 patients before results of a comparative study has been published.
Publication: Decision Support Document No. 93: https://eprints.aihta.at/1075/
Contact: Claudia Wild
Announcement
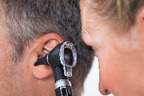
Balloon Eustachian Tuboplasty for the treatment of Eustachian tube dysfunction - German short version and update of the correspondent EUnetHTA assessment
Balloon Eustachian Tuboplasty (BET) is a minimally invasive treatment option for patients with chronic tube dysfunction which involves passing a balloon catheter into the Eustachian tube. Currently, there are 2 CE marked products on the market for BET: the Bielefelder Ballonkatheter/TubaVent® produced by Spiggle & Theis and the AERATM produced by Acclarent Inc./Johnson & Johnson.
This report, which is based on a EUnetHTA assessment published in February 2015, aims to compare the effectiveness and safety of BET in patients aged >12 years with tympanostomy and medication.
Publication: Decision Support Document No. 91: https://eprints.aihta.at/1073/
Contact: Julia Mayer

Newsletter
Announcement
Sponsoring of Patientinitiatives in Austria. Systematic Analysis
As part of the professionalization of patients organizations and the expansion of their activities (eg. awareness campaigns, advisory services for patients and their relatives, etc.) the need for financial resources is also increasing. This need is often covered by pharmaceutical and medical device manufacturers. This report analysed the extent of financial support for patient organizations from pharmaceutical companies in Austria. 24 of 115 PHARMIG member companies have declared donations to patient organization for the year 2014. A total of € 1,145.718.- grants were given. Most grants (63%) were given to four fields of diseases: neurology, haemato-oncology, rheumatology and haemophilia.
Publication: Rapid Assessment No 7b: https://eprints.aihta.at/1072
Contact: Claudia Wild
Project / HTA-methods & steering instruments
Involvement of the public and patients in HTA processes / programs International experiences and good practice examples
Project leaders: Claudia Wild
Project team: Andrea Fried
Duration: October 2015 – April 2016
Publikation: LBI-HTA Projektbericht Nr. 86: https://eprints.aihta.at/1088/
Announcement
Outpatient neurological rehabilitation: goal attainment in stroke patients. Systematic review.
In the Austrian rehabilitation plan aims for outpatient rehabilitation have been defined. These can be summarised as a higher flexibility of the therapy plan, a higher participation into the living environment, a higher activation of the self-help capability, a better re-integration into the work process and a better cooperation with aftercare units. Aim of this report (a master´s thesis) is to analyse, if outpatient stroke patients are able to achieve these goals. 7 observational pre-post studies (4 with control group) fulfilled the inclusion criteria. For the domains ‘functionality’ and ‘participation’ most of the studies found positive effects. The heterogeneity of instruments led to different results in the domain ‘satisfaction’. Positive trends could be observed for the domain ‘re-integration into the work process’. Finally, none of the studies reported results for the domain ‘aftercare’. Overall, the risk of bias of included studies has to be rated as high. We did not aim at comparing different rehabilitation settings. Future studies should aim at the further standardisation of instruments, the randomized allocation of patients to the rehabilitation settings and the investigation of patients’ expectations and needs.
Publication: Decision Support Document No. 92: https://eprints.aihta.at/1071
Contact: Brigitte Piso

Newsletter
Announcement
Horizon Scanning in Onkology - Reports
We are pleased to introduce our four new HSO reports.
DSD HSO No. 52
Carfilzomib (Kyprolis®) for patients with relapsed multiple myeloma who have received one to three prior lines of therapy
DSD HSO No. 53
Nivolumab (Nivolumab BMS®) for the second-line therapy of metastatic squamous non-small cell lung cancer
Project / HTA-methods & steering instruments
Project „Transparency“
Project leaders: Claudia Wild
Part 1: Sponsoring of continuing medical education (CME)
Part 2: Sponsoring of patient groups
- Update Sponsoring of patient initiatives in Ausrtria 2015 (2017)
- Update Sponsoring of patient initiatives in Austria 2016 (2017)
- Update Sponsoring of patient initiatives in Austria 2018 (2019)
- Update Sponsoring of patient initiatives in Austria 2019 (2021)
- Update Sponsoring of patient initiatives in Austria 2018 (2019)
- Update Sponsoring of patient initiatives in Austria 2021 (2023)
Part 3: Sponsoring of non-interventional (NIS) post-marketing studies
Part 4: Pharma: Disclosure of payments to doctors in Austria 2015
Project team:
Part 1: Roland Küllinger, Claudia Wild, Katharina Hintringer
Part 2: Ozren Sehic, Aamna Khan, Judit Erdos, Claudia Wild
Part 3: Nikolaus Gregor-Patera, Marlene Schader, Claudia Wild
Part 4: Stefanie Mantsch, Philipp Petersen, Claudia Wild
Announcement
Biodegradable stents for the treatment of refractory or recurrent benign oesophageal stenosis
Biodegradable ("resorbable") esophageal stents to treat benign refractory and/ or recurrent esophageal strictures were recently developed as alternatives to self-expanding metal stents (SEMS) and self-ex¬pandierenden plastic stent (SEPS). Only one product - the SX-ELLA stent Esophageal Degradable ™ BD - holds CE marking (since 2007) on the European market. Only a few studies of very low quality were available for the assessment of efficacy and safety. Currently, the biodegradable esophageal stents is an experimental procedure in an early stage of use.
Publication: Decision Support Document Nr. 89: https://eprints.aihta.at/1067/
Contact: Claudia Wild