News

Welcome to the homepage of AIHTA!
AIHTA is an academic non-profit institute. At our website we wish to present our research and give support for health care decision-making. Here all of our reports are available for free. For further information please contact the AIHTA team.
In the News section you will find our most recent publications: newsletter, project-reports etc. Previous reports you might find via Research areas, Research projects and Repository.
Announcement
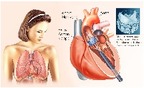
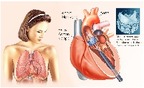
Percutaneous aortic valve replacement in Austria (Part II): Data analysis
A secondary analysis of reimbursement data (DRG-data) from public hospitals was conducted in order to determine how the Aortic Valve Replacement was implemented 2009-2016, either percutaneously (TAVI) or surgically (SAVR), in the Austrian healthcare system. For the period 2009 and 2016, in total 4,338 TAVI and 16,439 surgical aortic valve replacements (SAVR) were carried out. During this period the data show a constant rise in TAVI and a slight fall in SAVR use. The analysed data show a rate of 107 TAVI per million inhabitants in Austria.
Publication: LBI-HTA Project report No. 95b: https://eprints.aihta.at/1141/
Contact: Martin Robausch

Newsletter
Announcement
Horizon Scanning in Onkology - Reports
We are pleased to introduce our new HSO report.
DSD HSO No. 74
Midostaurin with standard chemotherapy in FLT3-Positive Acute Myeloid Leukaemia
Announcement
Transcatheter Aortic Valve Implantation Part I: A systematic review of health economic evaluations
For several years, the high-cost Transcatheter Aortic Valve Implantation (TAVI) has been a less invasive alternative to surgery for patients. The aim of the present work was to evaluate the health-economic evaluations of TAVI for inoperable patients and operable patients with high or intermediate surgical risk within a systematic review. The results showed that, TAVI was cost-effective compared to Medical Management in inoperable patients, whereas TAVI compared to Standard Management in inoperable patients and to Surgical Aortic Valve Replacement in patients with high surgical risk resulted in controversial results.
Publication: LBI-HTA Project report No. 95: https://eprints.aihta.at/1139
Contact: Sarah Wolf
Announcement
Horizon Scanning in Onkology - Reports
We are pleased to introduce our two new HSO reports.
DSD HSO No. 72
Abemaciclib (Verzenio®) in combination with fulvestrant for treatment of HR-positive, HER2-negative advanced breast cancer (ABC)
DSD HSO No. 71
Olaparib (Lynparza®) in patients with BRCA-mutated metastatic breast cancer

Newsletter
Event archive
Event
Symposium Post-marketing studies (application monitoring) in Germany and Europe - do they contribute to drug safety?
This Information is available in German only.

Newsletter
Announcement
Sponsoring of Patientinitiatives in Austria 2016. Update of the Systematic Analyses 2015 and 2014
As part of the professionalization of patients organizations and the expansion of their activities (eg. awareness campaigns, advisory services for patients and their relatives, etc.) the need for financial resources is also increasing. This need is often covered by pharmaceutical and medical device manufacturers. This update analyzed the extent of financial support for patient organizations from pharmaceutical companies in Austria in 2016. 40 of 118 PHARMIG member companies have declared donations to patient organization for the year 2016. A total sum of € 1.435.059 in grants to patient initiatives were reported by the PHARMIG member companies. Most grants (72%) were given to initiatives in the field of Oncology & Hematology, Neurology, Lung, Hemophilia, and Rheumatology.
Altogether the increase in disclosure in comparison to 2015 was much lower. The total sum of contributions even decreased by almost 20% compared to 2015.
Publication: Rapid Assessment Nr. 7b/2. Update 2017: https://eprints.aihta.at/1132
Contact: Philipp Petersen